On Apr. 28, 2026, FDA’s Commissioner Martin Makary and Chief AI Officer Jeremy Walsh announced an initiative to advance real-time clinical trials (RTCTs). This initiative leverages electronic health records and enables FDA reviewers to view safety signals and endpoints as trials progress. Two proof-of-concept RTCTs have been initiated.
FDA’s Deputy Chief Medical Officer Mallika Mundkur also announced a Request for Information (RFI) to shape the “AI-Enabled Optimization of Early-PhaseClinical Trials Pilot Program” launching this summer. Comments regarding pilot structures and evaluation metrics are accepted until May 29, 2026. For detailed RFI question descriptions, please visit Section II here. FDA intends to disseminate final selection criteria in July and complete pilot selections in August.
Goal: As a step toward “continuous” trials, FDA aims to explore how AI and data science can improve trial efficiency, enhance safety monitoring, facilitate dose selection decisions, and enable more informed early go/no-godecisions (e.g., a regulatory decision as to whether a Phase 1 study may proceed) while maintaining FDA’s rigorous scientific and regulatory standards and promoting trustworthy AI systems.
Proof-of-concept RTCTs:
- AstraZeneca is conducting a Phase 2 trial (TRAVERSE NCT05951959 acalabrutinib + venetoclax + rituximab) in treatment-naïve mantle cell lymphoma with participation from the University of Texas MD Anderson Cancer Center and the University of Pennsylvania. Paradigm Health provides the technical framework for real-time signal sharing.
- Amgen is conducting a Phase 1b trial (STREAM-SCLC with tarlatamab) in limited-stage small cell lung carcinoma (final site selection in process).
- For each trial, the FDA met with the sponsor on the establishment of criteria for reporting signals in real time.
- Both trials explore new uses of previously approved therapies.
Background:
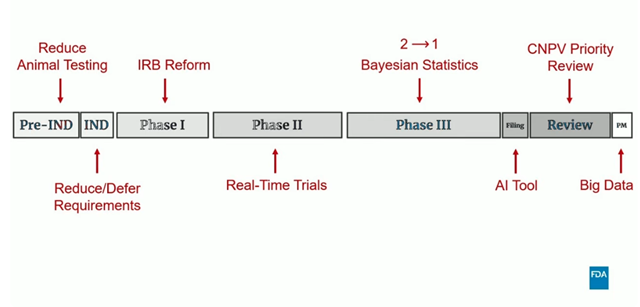
- FDA’s RTCT initiative is part of the broader effort accelerating drug development timelines and regulatory decisions (See figure below).
- The RTCT initiative involved FDA’s cross-center leadership and review divisions. Learnings may be applicable to pandemic preparedness.